Abstract: Niacinamide is more than a skincare buzzword; it is a clinically proven multitasker for the skin. Safely incorporated into a variety of cosmetic products, niacinamide acts almost as a skincare superhero. It can address wrinkles, reduce sebum production and inflammation, and improve the condition of acne, melasma and hyperpigmentation. Supported by clinical studies, this review explores the scientific evidence, demonstrating that niacinamide is more than a trend – it is a versatile ingredient for skin health and radiance.
Berglez P. Nurturing radiance: navigating the power of niacinamide. Cos ACTIVE J. 2025;3:22–29
WHAT IS NIACINAMIDE?
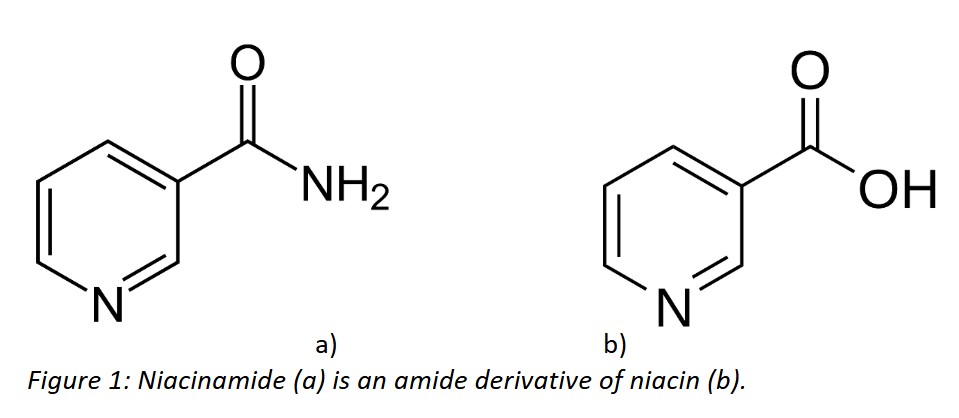
Niacinamide, also known as nicotinamide or vitamin B3, is an amide of niacin (Figure 1). It is a water-soluble substance of natural origin, also present in the human body, either in free or bound form, mainly as part of the coenzymes nicotinamide-adenine dinucleotide (NAD) and nicotinamide-adenine dinucleotide phosphate (NADP) (1). These coenzymes play an important role in cellular metabolism, in various metabolic pathways (2). NADP is involved in the biosynthesis of fatty acids, lipids and steroids, while NAD participates in cellular energy metabolism and energy production (3, 4).
Niacinamide is either synthesised in the body or obtained from foods such as meats, liver, yeast, dairy products, legumes, beans, nuts and seeds, green leafy vegetables, coffee and tea, or dietary supplements and medicines. Niacinamide is used in medicine primarily to treat niacin deficiency, because unlike nicotinic acid, it does not cause flushing, itching, burning sensation or tingling after oral application. In recent years, it has also become a hot topic in the cosmetics world due to its beneficial effects on the skin (5‒7).
MOLECULAR INSIGHTS RELEVANT FOR FORMULATING
Although niacinamide occurs in nature, for cosmetic or medicinal use it is synthesised from either nicotinic acid or 3-cyanopyridine (8–10). It appears as an odourless, colourless, crystalline solid or as a white crystalline powder with a bitter taste (11–13).
Niacinamide dissolves readily in water (about 690 g/L at 20 °C), and is also soluble in glycerol (0.1 g/mL), ethanol (660 g/L at 20 °C) and propylene glycol (28 g/100 mL at 32 °C) (14–16).
Niacinamide hydrolyses to niacin under strongly alkaline or acidic conditions, and the rate of hydrolysis depends on pH. For example, it takes 15 hours for half of the niacinamide to hydrolyse at pH 9.05, 45 hours at pH 1.38, 85 hours at pH 2.03 and 115 hours at pH 8.41. Hydrolysis is minimal at pH values from 4.5 to 6. In this range, at 32 °C, the half-life of niacinamide is about 1000 days. Niacinamide is also heat stable; a 10% solution can be heated to 120 °C for 20 minutes without detectable niacin formation (17).
NIACINAMIDE AS A COSMETIC INGREDIENT WITH A FOCUS ON SAFETY
Niacinamide is a versatile cosmetic ingredient. It is used in a variety of products, from body, hand and facial creams, serums, lotions, powders, sprays and tonics, to skin cleansers, shampoos and conditioners. The concentrations of niacinamide in products on the market range from as low as 0.0001% to as high as 100%, but are usually from 3 to 5%. Concentrations of up to 10% did not produce stinging in clinical testing. Formulations containing niacinamide did not cause significant adverse skin reactions such as irritation, sensitisation or photosensitisation. While some tested niacinamide formulations were found to be mild ocular irritants, others were not (18). It is worth noting that niacinamide is also classified as GRAS (generally recognised as safe) (19).
CLINICAL EXPLORATION
A number of clinical studies have demonstrated the effectiveness of niacinamide in various skin conditions, for both cosmetic and dermatological applications, whether used alone or compared with other active ingredients.
Before we delve into clinical studies, it should be clearly emphasised that, while clinical studies provide valuable insight into niacinamide’s cosmetic activity, such as effective concentration ranges, expected skin benefits and potential for irritation or other adverse skin reactions, they must be used responsibly in formulation and marketing. Cosmetic products cannot make medical claims, and formulations should not be directly linked to specific clinical studies, even when using similar concentrations. Each study is conducted on a particular product with its own composition and variables, which cannot simply be equated with ours. Responsible communication means being honest, transparent and fair to consumers.
Acne
A 4% niacinamide gel, compared to a 1% clindamycin gel, demonstrated similar effectiveness in treating moderate inflammatory acne vulgaris. In this randomised, double-blind clinical study by Khodaeiani et al., 40 patients in each group were further divided into subgroups based on skin type (oily or non-oily). The grade of facial acne and the total number of facial papules and pustules were recorded at each visit. The results showed that niacinamide gel was more effective on oily skin than on non-oily skin when compared to clindamycin gel. No major skin reactions were observed during the eight-week study period with twice-daily application. While both active ingredients showed similar effectiveness in treating acne, a notable advantage of niacinamide over clindamycin is the absence of risk for the development of antibiotic resistance (20). An earlier, similarly designed study by Shalita et al., involving 76 patients, showed a comparable but significant reduction in acne in both groups, with reductions of 82% and 62% for niacinamide and clindamycin, respectively (21).
In an outpatient setting, 38 patients aged 18 to 25 years with mild to moderate acne applied a 4% niacinamide gel for eight weeks. The treatment resulted in a significant reduction in pustules, papules and comedones. Some side effects occurred, including one case of pruritus and three cases of mild burning sensation (22).
Two studies by the same researchers (Draelos et al.) compared a 2% niacinamide and an 1% D‐panthenol gel with a placebo in reducing the sebum excretion rate. The first study was a four-week, double‐blind, placebo‐controlled study involving 100 Japanese women, while the second was a six-week, randomised, split‐face study involving 30 Caucasian women. Participants in both studies were aged between 20 and 49 years. In the first study, measurements in the niacinamide group showed a reduction in sebum excretion rate of approximately 21%. The results of the second study did not fully align with those of the first, as sebum excretion rate was not reduced, although the niacinamide group did show lower sebum levels. The authors speculate that the discrepancy in results may be due to the split-face study design and possible cross-contamination with the placebo side of the face (23).
Other dermatological skin conditions
A prospective, randomised, double-blind, placebo-controlled clinical study was conducted on 60 subjects aged 18 to 65 years with diagnosed discoid lupus erythematosus, an autoimmune connective tissue disorder. Patients were assigned to 2%, 4% or placebo groups, and received a cream for facial lesions or a gel for scalp lesions, applied topically twice daily for 12 weeks. Results showed that dermal use of niacinamide as an adjuvant to other treatment regimens demonstrated good cosmetic outcomes and minimal side effects. Dermal 4% niacinamide preparations were more effective on lesions than the 2% gel and cream, although more cases of irritation occurred at the higher concentration (24).
36 males and 12 females aged 20 to 50 years with mild to moderate seborrhoeic dermatitis were treated once daily for 12 weeks with either a 4% niacinamide cream or a placebo cream. Erythema, scaling, infiltration and adverse reactions were assessed before starting treatment and after 2, 6 and 12 weeks of treatment. A significant 75% reduction in predefined scores for erythema, scaling and infiltration was observed compared to baseline for the niacinamide cream, and 35% for the placebo. Although adverse events occurred in two patients using the niacinamide cream, these included only minimal burning sensation and pruritus (25).

Cosmetic antiaging applications
A double-blind, split-face, randomised, controlled study was conducted on 30 healthy Japanese women aged 31 to 49 years with wrinkles in the eye area. The volunteers applied a 4% niacinamide cream to one side of the face and a vehicle cream to the other side daily for eight weeks. Significant improvement in skin wrinkles and texture was observed in 64% of volunteers compared to the control. The cream was generally well tolerated; only minimal irritation occurred in one case (26).
A similar study involving 50 Caucasian women aged 40 to 60 years was conducted over 12 weeks. The volunteers applied a 5% niacinamide cream twice daily to one side of their face and a placebo to the other side. Overall skin improvement was observed, with prevention of skin yellowing and a significant reduction in fine lines and wrinkles (27).
In a double-blind, placebo-controlled, split-face study, 27 women with melasma, aged 25 to 53 years, applied a 4% niacinamide cream to one side of the face and a 4% hydroquinone cream to the other side for eight weeks. The women used sunscreen during the study. Improvements in melasma were observed on both sides of the face, with no statistically significant difference in colorimetric measurements. Mild adverse effects were observed on the niacinamide-treated side (18% of patients experienced erythema, pruritus or burning sensation), compared to moderate adverse effects on the hydroquinone-treated side (29% of patients). Good-to-excellent improvement was observed in 44% of patients using niacinamide and 55% of patients using hydroquinone. Post-treatment biopsies showed that niacinamide improved solar elastosis and resulted in a significantly lower inflammatory infiltrate. It is worth noting that the skin lightening effect of niacinamide became evident later in the treatment than that of hydroquinone. Researchers concluded that the lower incidence of adverse effects with niacinamide suggests it could be used for longer periods or as a maintenance cosmetic ingredient (28).
Two clinical studies by Hakozaki et al, with different study designs, were conducted to assess the reduction in pigmentation and suppression of melanosome transfer with 2% and 5% niacinamide moisturisers. The 5% test group, in a randomised, split-face, double-blind, paired design, included 18 Japanese women aged 25 to 60 years with hyperpigmentation, who applied a niacinamide moisturiser to one side of the face and a vehicle moisturiser to the other side twice daily for eight weeks. After four weeks, the niacinamide moisturiser significantly decreased hyperpigmentation and increased skin lightness compared to the control. However, a plateau in treatment effect was observed after four weeks. The study did not mention any adverse effects (29).
The 2% test group, in a randomised, split-face, double-blind, round-robin design, comprised of 120 Japanese women aged 18 to 30 years with moderate to deep facial tan. In subgroups of 40, they used two of three treatments: a vehicle moisturiser, a sunscreen (SPF 15), and a 2% niacinamide moisturiser with sunscreen, twice daily for eight weeks. The difference in skin lightening effect between the niacinamide formulation with sunscreen and sunscreen alone was similar to the difference between sunscreen and the control vehicle. It was concluded that adding 2% niacinamide to an SPF 15 sunscreen provided an additional skin lightening effect comparable to that achieved by the SPF 15 sunscreen alone (29).
The in vitro data obtained in the same study (29) suggest that niacinamide is efficient at skin lightening by inhibiting melanosome transfer from melanocytes to keratinocytes, rather than by directly influencing melanin synthesis in melanocytes.
In addition to skin lightening, niacinamide also affects several other epidermal parameters. A study on the forearms of 20 healthy volunteers aged 21 to 28 years, who applied a vehicle containing 5% (w/v) niacinamide twice daily for 28 days, showed significantly lower transepidermal water loss values and an increase in corneocyte size and maturity compared to areas treated with the vehicle alone. A test formulation with niacinamide significantly increased epidermal thickness by almost 10%. Niacinamide also exhibited anti-inflammatory action (30).
Niacinamide can also be combined with other cosmetically active ingredients, such as peptides, antioxidants and retinyl propionate, to achieve wrinkle reduction (31).
In summary, the clinical studies described demonstrate the benefits of niacinamide in skincare and dermatology. Scientific evidence supports the diverse effects of niacinamide on the skin, from improvements in hyperpigmentation, melasma, wrinkles, acne, sebum production and inflammation.

EVIDENCE-BASED FORMULATING
As shown in the previous clinical review section, niacinamide concentrations of 2 to 5% had positive effects on skin conditions such as acne, dermatitis, hyperpigmentation and wrinkles.
In a recent study of 15 cosmetic products available on the market, the concentration of niacinamide ranged from 0.030% to 11.217% (w/w). Most products (40%) were within the 2.0–2.5% (w/w) range, followed by 4.2–4.6% (w/w; four products) and 10.1–11.2% (w/w; three products). Chemical analysis of four products with declared niacinamide concentrations showed that one product contained 101.8% of the declared concentration and another 112.2%, whereas the remaining two contained only 46.9% and 56.5% of the concentrations declared on the label (32).
There is no upper limit on niacinamide concentration in cosmetics according to the EU cosmetic Regulation (EC) No 1223/2009. Currently, at least one cosmetic product on the market contains 100% niacinamide; this product is advised to be mixed with a suitable base (33). In this context, it should be stressed that the effects of higher niacinamide concentrations on the skin have not yet been investigated. However, clinical studies confirm that niacinamide, when used in typical concentrations, is a non-irritant and is neither sensitising nor photosensitising (18).
As previously noted, niacinamide has good water solubility. When incorporated into formulations, it is most stable within a pH range of 4.5 to 6, but hydrolyses to niacin under more alkaline or acidic conditions. Additionally, niacinamide is heat stable.
In aqueous solution, a combination of niacinamide and ascorbic acid tends to form a yellow colour, attributed to a 1:1 niacinamide and ascorbic acid complex. This association is pH-dependent and peaks at pH 3.8. It has been hypothesised that this occurs due to a charge-transfer interaction between protonated nicotinamide and ascorbate ion. The complex formation is reversible, and if the formulation pH differs from 3.8, the extent of complex formation is reduced (34).
This does not, however, mean that the complex is completely ineffective on the skin. In an ex vivo experiment on human skin, explants were treated with an O/W emulsion containing the niacinamide-ascorbic acid complex and later exposed to visible light, UVA and UVB rays. The results indicate a possible absence of melanin transfer in vivo, but further tests would be needed to confirm this hypothesis (35).
In vitro studies using human and porcine epidermis and artificial membrane were performed to investigate the enhancement of niacinamide skin penetration, comparing the permeation of niacinamide in different solvents, as well as in binary and ternary mixtures, under dose conditions of 5 μL/cm2. Niacinamide in a binary mixture of propylene glycol with oleic acid at a ratio of 10:90, and propylene glycol with linolenic acid in a ratio of 50:50, achieved higher skin penetration compared with propylene glycol alone. These results confirm that propylene glycol alone and specific mixtures with fatty acids can act as permeation enhancers, resulting in improved permeation of niacinamide (36). It should be noted that propylene glycol is generally not accepted within the concept of natural cosmetics, as defined by the criteria of certification institutions.
The key findings were similar in an in vivo human study of niacinamide-enhanced dermal delivery involving six healthy volunteers (37). It has been hypothesised that oleic acid creates permeable defects in the stratum corneum, which consequently increase the diffusion of propylene glycol with dissolved niacinamide (36).
In the future, further scientific research on niacinamide is expected to focus on developing new nanotechnology vehicles such as microemulsions, microneedle-like particles and multiple emulsions to improve dermal penetration, delivery and photoprotection (38–40).
CONCLUSION
Niacinamide is an excellent cosmetic ingredient with numerous beneficial effects on the skin. It can be applied to acne-prone skin, reducing sebum production and inflammation, two key factors in acne. In addition to addressing concerns related to ageing skin, such as reducing fine lines, wrinkles, hyperpigmentation and melasma, niacinamide also improves skin elasticity. It may cause mild and rare adverse effects. It is a safe and stable cosmetic ingredient in a wide range of concentrations. Niacinamide can be used alongside traditional acne medications, with fewer side effects and no risk of bacterial resistance. It offers considerable diversity in formulation possibilities and holds significant potential for future use and new skin delivery options.
Tch. Assist. Pia Berglez, B. Sc. Cosmetic Science, M. Ind. Pharm.
University of Ljubljana, Faculty of Pharmacy, Ljubljana, Slovenia
pia.berglez@ffa.uni-lj.si

References
Please click on the references below for more information.
11. Van Arnum SD. Niacin, nicotinamide, and nicotinic acid. In Kirk RE, Othmer DF, Grayson M et al., editors. Encyclopedia of chemical technology Vol. 25. New York, NY: John Wiley & Sons.; 2004. p. 84.
12. Osol A et al. Remington’s Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co.; 1975. 950 p.
13. Larranaga MD, Lewis RJ Sr., Lewis RA. Hawley’s Condensed Chemical Dictionary. 16th ed. Hoboken: John Wiley & Sons, Inc.; 2016. 966 p.

Leave a Reply